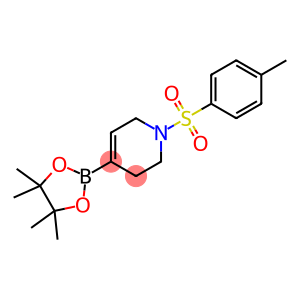
1-Tosyl-1,2,3,6-tetrahydropyridine-4-boronic acid, pinacol ester
1-[(4-Methylphenyl)sulfonyl]-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1,2,3,6-tetrahydropyridine
CAS: 1256360-46-9
Molecular Formula: C18H26BNO4S
1-Tosyl-1,2,3,6-tetrahydropyridine-4-boronic acid, pinacol ester - Names and Identifiers
1-Tosyl-1,2,3,6-tetrahydropyridine-4-boronic acid, pinacol ester - Physico-chemical Properties
| Molecular Formula | C18H26BNO4S |
| Molar Mass | 363.28 |
| Density | 1.2±0.1 g/cm3 |
| Boling Point | 444.2±55.0 °C at 760 mmHg |
| Flash Point | 222.5±31.5 °C |
| Vapor Presure | 0.0±1.1 mmHg at 25°C |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Sensitive | IRRITANT |
| Refractive Index | 1.558 |
| MDL | MFCD17926518 |
1-Tosyl-1,2,3,6-tetrahydropyridine-4-boronic acid, pinacol ester - Introduction
4-(4,4, 5,5-tetramethyl-[1,3,2] dioxaborolan-2-yl)-1-(toluene-4-sulfonyl)-1,2,3,6-tetrahydropyridine is an organic compound with a variety of properties and uses.
Nature:
-Appearance: A white to off-white crystalline solid.
-Melting point: about 125-130°C.
-Solubility: Soluble in common organic solvents (such as methanol, ethanol, chloroform, etc.), but insoluble in water.
-Stability: relatively stable under conventional conditions, but can be decomposed or discolored by heat.
-Chemical properties: As a derivative of pyridine, it has similar chemical reactivity to pyridine.
Use:
-Catalyst: This compound can be used as a precursor of a coordination compound catalyst to participate in various organic synthesis reactions, such as hydrogenation reactions, arylene alkene reactions, etc.
-Research field: Due to its special structure and reaction properties, the compound is widely used in the fields of organic synthesis, coordination chemistry and materials science.
Method:
The synthesis method of the compound is relatively complex, generally using multi-step reaction, including ring reaction, substitution reaction, coordination reaction and so on.
Safety Information:
-The compound is not specifically classified as a hazardous substance, but it is still necessary to pay attention to safe handling measures, such as wearing protective gloves, safety glasses, etc.
-Avoid contact and inhalation of the compound and avoid swallowing.
-Avoid contact of the compound with skin, eyes and mucous membranes. If contact occurs, rinse immediately with plenty of water and seek medical help.
-storage should be sealed, stored in a dark, dry place.
Nature:
-Appearance: A white to off-white crystalline solid.
-Melting point: about 125-130°C.
-Solubility: Soluble in common organic solvents (such as methanol, ethanol, chloroform, etc.), but insoluble in water.
-Stability: relatively stable under conventional conditions, but can be decomposed or discolored by heat.
-Chemical properties: As a derivative of pyridine, it has similar chemical reactivity to pyridine.
Use:
-Catalyst: This compound can be used as a precursor of a coordination compound catalyst to participate in various organic synthesis reactions, such as hydrogenation reactions, arylene alkene reactions, etc.
-Research field: Due to its special structure and reaction properties, the compound is widely used in the fields of organic synthesis, coordination chemistry and materials science.
Method:
The synthesis method of the compound is relatively complex, generally using multi-step reaction, including ring reaction, substitution reaction, coordination reaction and so on.
Safety Information:
-The compound is not specifically classified as a hazardous substance, but it is still necessary to pay attention to safe handling measures, such as wearing protective gloves, safety glasses, etc.
-Avoid contact and inhalation of the compound and avoid swallowing.
-Avoid contact of the compound with skin, eyes and mucous membranes. If contact occurs, rinse immediately with plenty of water and seek medical help.
-storage should be sealed, stored in a dark, dry place.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-tosyl-1,2,3,6-tetrahydropyridine Visit Supplier Webpage Request for quotationCAS: 1256360-46-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-tosyl-1,2,3,6-tetrahydropyridine Request for quotation
CAS: 1256360-46-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1256360-46-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-tosyl-1,2,3,6-tetrahydropyridine Visit Supplier Webpage Request for quotation
CAS: 1256360-46-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1256360-46-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-tosyl-1,2,3,6-tetrahydropyridine Visit Supplier Webpage Request for quotationCAS: 1256360-46-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-tosyl-1,2,3,6-tetrahydropyridine Request for quotation
CAS: 1256360-46-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1256360-46-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-tosyl-1,2,3,6-tetrahydropyridine Visit Supplier Webpage Request for quotation
CAS: 1256360-46-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1256360-46-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


